Keywords
stroke, digital health, discrete choice experiments, scoping review, preferences, digital health technologies
Digital Health Interventions (DHIs) are being increasingly incorporated into stroke prevention, treatment, and long-term rehabilitation, offering promising solutions to improve access, efficiency, and outcomes in stroke care. Understanding users’ preferences is essential to ensure the relevance and adoption of the technologies. Discrete Choice Experiments (DCEs) have emerged as a widely used survey-based method for eliciting preferences, enabling the quantification and ranking of healthcare-related choices of stakeholders. However, there is a knowledge gap about the extent to which DCEs have been used to evaluate DHIs in stroke care. Hence, this scoping review aimed to map the existing literature on this topic and examine the use of the DCE methodology to elicit preferences for DHIs in stroke care.
The review followed the JBI methodology for scoping reviews. Reporting was guided by the Preferred Reporting Items for Systematic Reviews and Meta-analysis extension for Scoping Reviews. A comprehensive search was conducted across four databases: Embase, PubMed, Scopus, and Web of Science without restrictions on publication date. Eligible studies included those employing DCEs to evaluate preferences for DHIs in stroke care. The terms identifying “stroke”, “discrete choice experiment”, and “digital health intervention/digital health technology” were combined in the search string. Retrieved studies were screened and analysed by two reviewers independently. Conflicts were resolved by team consensus.
A total of 1,846 studies were identified. After removing duplicates (n = 442), 1,404 titles and abstracts were screened. Thirteen full-text studies were assessed for eligibility, but none satisfied the inclusion criteria of the study.
The scoping review did not identify any study that applied DCE methods to evaluate preferences for DHIs in stroke care. Future research should consider DCE methodologies in evaluating preferences for DHIs to inform the design, adoption, and implementation of digital health solutions in stroke care.
stroke, digital health, discrete choice experiments, scoping review, preferences, digital health technologies
Stroke is a leading cause of long-term disability worldwide, with many survivors requiring ongoing support to manage the physical and cognitive effects (Elendu et al., 2023). Estimates from The Global Stroke Fact Sheet 2025 reported approximately 94 million cases of prevalent stroke and 11.9 million cases of incident strokes worldwide (Feigin et al., 2025). Up to 74% of stroke survivors require some assistance or rely on caregiver support to continue with activities of daily living (Miller et al., 2010). Over the period from 1990 to 2021, the incidence of stroke increased markedly, while the global economic burden of stroke is estimated to be approximately US$890 billion (Mozaffarian et al., 2016). Stroke exerts a substantial burden on health, often leading to a wide range of complications. These include myocardial infarction, cardiac arrhythmia, heart failure, pneumonia, dysphagia, intestinal bleeding, urinary tract infections, urinary incontinence, hip fractures, and pain, among others (Kumar et al., 2010).
Given the substantial health and economic burden of stroke, Digital Health Interventions (DHIs) offer a potential means by which to support stroke care and rehabilitation (Schwamm & Silva, 2023). Digital Health Technologies (DHTs) have expanded considerably in healthcare over the past two decades, especially during the COVID-19 pandemic (Vo et al., 2024), when lockdowns and mandatory social distancing limited in-person access to healthcare. In the literature, “Digital Health Intervention (DHI)” and “Digital Health Technology (DHT)” are terms used interchangeably (Mandyam et al., 2025). DHIs are defined as “the use of information and communications technologies in medicine and other health professions to manage illnesses and health risks and to promote wellness” (Ronquillo et al., 2025). They include a broad range of applications, such as telemedicine, wearable technologies, mobile health (mHealth), electronic medical records (EMRs), remote monitoring, and artificial intelligence-supported systems. DHIs enhance healthcare delivery and improve clinical outcomes by extending care beyond traditional clinical settings and enabling chronic disease management with fewer in-person visits and shorter hospital stays (Golinelli et al., 2020; Krick et al., 2019).
The successful implementation of DHIs requires both clinical effectiveness and the ability to meet users’ needs and preferences (Mair et al., 2025; Or et al., 2011; Wilson et al., 2021). Effective digital solutions can fail to reach full potential if users are unwilling to adopt them, a phenomenon that has been observed especially among older populations (Deidda et al., 2018; Greenhalgh et al., 2017). Common barriers to adoption include concerns about usability, data privacy and security, cost, digital literacy, and trust (Krick et al., 2019; Or et al., 2011). In addition, insufficient training, particularly among healthcare professionals, hinders implementation, especially in cases that require problem-solving and communication skills (Alotaibi et al., 2025).
In this context, the discrete choice experiment (DCE) methodology may provide a useful means of understanding the facilitators and barriers to DHI adoption amongst stroke patients. DCE is a particular type of stated preference method for evaluating individual preferences (Clark et al., 2014). The basic premise of a DCE is that a hypothetical product or service, such as a DHI, can be described based on a number of defining characteristics, referred to as attributes, and individuals assess the desirability of the product or service on the basis of these attributes (Friedel et al., 2022; Ryan et al., 2001; Szinay et al., 2021). They present hypothetical scenarios in the form of choice tasks defined by attributes and their corresponding levels (Ryan, 2004). Table 1 presents an example of a choice task designed to elicit preferences for treating sarcopenia. Respondents are asked to choose between prioritising treatment for two different patients, each with distinct combinations of attributes (in the first column) and levels (shown in the corresponding rows).
Table 1 presents an example of a choice task designed to elicit preferences for treating sarcopenia. The choice task was extracted from Hiligsmann et al. (2020). Respondents are asked to prioritise treatment between two different patients, each with distinct combinations of attributes and levels. The first column of the table lists attributes, and the corresponding rows show the levels.
| Which of the patients do you think deserves treatment the most? (Tick one box only) | Patient A | Patient B |
Choices made in DCEs generate a preference-based ranking of the importance of different attributes and their corresponding levels. This information can inform the implementation of strategies which satisfy the needs and requirements of the target population. Analysis of DCE data also enables the exploration of preference heterogeneity, both at the individual and group levels. For example, it can be possible to examine differences in preferences based on observable respondent characteristics, such as rural versus urban, vulnerable population in comparison with the general population, and socioeconomic status (Lancsar & Louviere, 2008).
The development of DCEs needs to follow guidelines for good research practices, such as those outlined by ISPOR—The Professional Society for Health Economics and Outcomes Research (Bridges et al., 2011). These guidelines require researchers to establish and clearly define relevant attributes and their associated levels, as well as to develop a clear plan for analysis methods to be used (Hauber et al., 2016). Attributes and levels can be identified through several approaches (Bohorquez et al., 2024), including conducting literature searches relevant to the topic. Additionally, focus group discussions with patients, healthcare professionals, and other relevant stakeholders enable researchers to gain a deeper understanding of the attributes that matter most to these groups of participants. Think-aloud sessions, interviews, and consultations with subject matter experts are also viable options (Ryan et al., 2009).
DCE data are analysed using regression models (such as mixed logit or latent class) to estimate the relative importance of each attribute and the trade-offs individuals are willing to make (Lancsar et al., 2017). Although DCEs yield meaningful preference data, they introduce several methodological hurdles (Norman et al., 2019). From a researcher’s point of view, designing choice tasks involves selecting alternatives that are both policy-relevant and realistic, and describing them in a way that every respondent interprets them identically. On the participant side, even a well-constructed DCE can be overwhelming. Presenting too many attributes, levels, or repeated choice tasks can fatigue respondents and lead to inconsistent decision-making. This cognitive burden is especially pronounced among groups with limited experience in survey research or those with diseases that affect cognitive abilities, potentially leading to higher dropout rates or inconsistent responses (Ellis, 2021).
Despite these challenges, DCEs are widely used to measure health-related preferences and, in the context of this study, have been used to identify factors influencing patient preferences for telerehabilitation compared to in-person physiotherapy (Ross et al., 2025), estimate willingness to pay for telerehabilitation among people with chronic knee pain (Woode et al., 2025) and investigate preferences of individuals with chronic kidney disease towards wearable DHTs (Gc et al., 2022). While previous studies have reviewed DCEs in stroke care (Jolliffe et al., 2024), we did not come across any that focused on DHIs in stroke care, implying a lack of consolidated evidence synthesising information about the application of DCE to elicit preferences for DHIs in stroke care.
This scoping review, therefore, aimed to map the existing literature on this topic and examine the use of the DCE methodology to evaluate preferences for DHIs in stroke care. Specifically, the review sought to identify the participants involved in the DCEs, determine the DHIs assessed, identify the attributes and levels, and describe how these were selected for inclusion in the DCEs. Additionally, the review sought to identify the preferences and trade-offs reported, as well as the statistical approaches used.
Understanding how these studies were conducted allows for the identification of gaps for further exploration and method development. Systematically evaluating the components of the study design, such as participant selection and preparatory work related to attributes, levels, and choice tasks, may facilitate the application of similar methods in other contexts. Additionally, recognising the importance of Public and Patient Involvement (PPI) in research, the impact of stroke on cognition, and the cognitive demands associated with DCE study methodologies, made this study timely and relevant to inform future DCEs for DHIs in stroke care.
This scoping review followed the JBI methodology for scoping reviews, which entails 9 steps: (1) defining and aligning objective(s) and question(s); (2) developing and aligning the inclusion criteria with the objective(s) and question(s); (3) describing the planned approach to evidence searching, selection, data extraction, and presentation of the evidence; (4) searching for the evidence; (5) selecting the evidence; (6) extracting the evidence; (7) analysis of the evidence; (8) presentation of the results; and (9) summarising the evidence in relation to the purpose of the review, making conclusions and discussing the implications of the findings (Peters et al., 2024). In this review, the methods section explains steps 1 to 7, while steps 8 and 9 are detailed in the results and discussion sections, respectively. Furthermore, the reporting of this review is guided by the checklist of the Preferred Reporting Items for Systematic Reviews and Meta-analysis extension for Scoping Reviews (PRISMA-ScR) (Tricco et al., 2018).
The development of the review title, objectives and research questions was reported in the review protocol, which was registered on the Open Science Framework (OSF) on 2nd September 2025 (Forde et al., 2025a). The review examined the application of DCE methodology to evaluate DHIs in stroke care by exploring the following research questions: (1) Have DCEs been used to elicit preferences for DHIs in stroke studies? (2) What methodological approaches have been adopted?
The Population, Concept and Context (PCC) (Peters et al., 2024) was adopted to guide the inclusion and exclusion criteria of the review. The review considered studies conducted in the stroke disease area, regardless of whether the studies targeted patients, their families, friends, neighbours, caregivers, healthcare professionals, or representatives of the general population, among other stakeholders. The concept was the methodological conduct of DCEs to elicit preferences, while the context under consideration was DHIs in stroke care.
Original studies employing DCE methodology, whether based on online, paper-based, or interview surveys, were considered. In contrast, qualitative studies, systematic reviews, other types of reviews, commentaries, editorial letters, text and opinion papers, protocols, conference abstracts and proceedings were excluded.
The planned approach to carry out the different stages of the scoping review, including evidence searching, evidence selection, data extraction, and data presentation and charting, was recorded in the registered review protocol on the Open Science Framework (OSF).
The reviewers collaborated with an Information Specialist from RCSI University of Medicine and Health Sciences library to develop the search strategy using the Peer Review of Electronic Search Strategies (PRESS) elements. The search strategy was built on three keywords: “stroke”, “discrete choice experiment”, and “digital health intervention/digital health technology”. The PubMed search strategy and results are presented in Table 2. While search strategies of previous reviews on DCEs included Conjoint Analysis (CA) terms (Soekhai et al., 2019), our review did not include such terms since these are two distinct methodologies built on two distinct theories (Louviere et al., 2010)
Table 2 presents the search strategy used in PubMed. The table presents the date on which the search was conducted and the keywords that were used. It also presents the number of articles that each search string yielded, as well as the number of articles yielded after applying filters.
| PubMed search conducted on 03/09/2025 | ||
|---|---|---|
| Stroke keywords | Results | |
| 1 | "Stroke"[Mesh] OR "Stroke*"[Title/Abstract] OR "Apoplexy"[Title/Abstract] OR "CVA"[Title/Abstract] OR "Cerebrovascular Accident*"[Title/Abstract] OR "Cerebral Stroke"[Title/Abstract] OR "Cerebrovascular Accident*"[Title/Abstract] OR "Cerebrovascular Accident, Acute"[Title/Abstract] OR "Cerebrovascular Apoplexy"[Title/Abstract] OR "Cerebrovascular Stroke*"[Title/Abstract] OR "Stroke, Acute"[Title/Abstract] OR "Vascular Accident, Brain"[Title/Abstract] OR "Cerebrovascular Disorder*"[Title/Abstract] OR "Intracranial Arteriosclerosis"[Title/Abstract] OR "Intracranial Embolism and Thrombosis"[Title/Abstract] OR "Hemorrhagic Stroke*"[Title/Abstract] OR "Haemorrhagic Stroke*"[Title/Abstract] OR "Embolic Stroke*"[Title/Abstract] OR "Thrombotic Stroke*"[Title/Abstract] OR "cerebrovascular insult*"[Title/Abstract] OR "CVI"[Title/Abstract] OR "Cerebrovascular lesion*"[Title/Abstract] OR "CVL"[Title/Abstract] OR "brain attack*"[Title/Abstract] (Mphepo et al., 2025) | 432,101 |
| Discrete Choice Experiment keywords | ||
| 2 | "discrete choice experiment"[tiab] OR "choice analysis"[tiab] OR "choice method*"[tiab] OR "stated preference"[tiab] OR "discrete choice"[tiab] OR "discret*"[tiab] OR "willingness-to-pay"[tiab] OR "willingness to pay"[tiab] OR "willingness-to-accept"[tiab] OR "willingness to accept"[tiab] OR preference*[tiab] OR "discrete-choice"[tiab] OR "pairwise choice"[tiab] OR "Matched Pair Analysis"[tiab] OR "Matched-Pair Analysis"[tiab] OR "choice survey*"[tiab] OR "choice set*"[tiab]OR choice task*[tiab] | 373,251 |
| Digital Health Intervention/Digital Health Technology keywords | ||
| 3 | "digital health"[tiab] OR "digital health technolog*"[tiab] OR "digital health intervention"[tiab] OR "digital therap*"[tiab] OR "mobile application"[tiab] OR "mobile applications"[tiab] OR "mobile app"[tiab] OR "mobile apps"[tiab] OR "smartphone apps"[tiab] OR "smartphone app"[tiab] OR "cellphone app"[tiab] OR cellphone*[tiab] OR "cell phone*"[tiab] OR "cell phone app"[tiab] OR "mobile phone"[tiab] OR telehealth[tiab] OR telemedicine[tiab] OR telerehabilitation[tiab] OR telecare[tiab] OR "virtual medicine"[tiab] OR "virtual care"[tiab] OR "mobile health"[tiab] OR "mhealth"[tiab] OR "m-health"[tiab] OR "ehealth"[tiab] OR "e-health"[tiab] OR "electronic health"[tiab] OR "virtual consultation"[tiab] OR "computer-aided"[tiab] OR "computer aided"[tiab] OR "computer-assisted"[tiab] OR "computer assisted"[tiab] OR "remote monitoring"[tiab] OR "artificial intelligence"[tiab] OR AI[tiab] OR telestroke[tiab] OR "tele-stroke"[tiab] OR "mobile stroke"[tiab] OR "m-stroke"[tiab] OR mstroke[tiab] OR "virtual reality"[tiab] OR vr[tiab] OR "vr-aided"[tiab] OR "vr aided"[tiab] OR "vr-assisted"[tiab] OR "vr assisted"[tiab] OR "augmented reality"[tiab] OR wearable*[tiab] OR software*[tiab] OR device*[tiab] OR app[tiab] OR application*[tiab] OR android[tiab] OR "iOS"[tiab] OR tablet*[tiab] OR smartwatch[tiab] OR "smart watch"[tiab] OR "smart technolog*"[tiab] OR "smart tech*"[tiab] OR "hands free"[tiab] OR "hands-free"[tiab] OR wireless[tiab] OR "wire-less"[tiab] OR "wire less"[tiab] OR voice[tiab] OR "voice command"[tiab] OR "voice control"[tiab] OR portable[tiab] | 3,156,745 |
| 4 | #1 AND #2 AND #3 | 540 |
| Filters applied | ||
| English, Full-text | 510 | |
The identification of relevant studies was conducted in two stages. A preliminary search was conducted on PubMed to identify primary studies on the topic, and possible keywords and descriptions used in the titles and abstracts to expand the search. The search strategy was then developed according to the specificity of each of the included databases: Embase, PubMed, Scopus and Web of Science. The PubMed search filtered articles published in English language and in full text, whereas the Embase search applied the English language filter only. No filters were applied to the search results of Scopus and Web of Science. The final study search across all databases was conducted on 3rd September 2025.
All the retrieved references were imported into Covidence, a software for managing reviews, where most of the duplicates were automatically removed (Veritas Health Innovation, 2025). Two reviewers (FF and TM) independently conducted the title and abstract screening as well as the full-text screening according to the inclusion and exclusion criteria. Conflicts that emerged at each stage of the review were adjudicated by a third, independent reviewer (SS) in consultation with the team.
A newly developed data extraction template was created by the review team based on the objectives of the review. The content of the data extraction included: study identification information (study ID, journal name, study title, year of publication, country where the study was conducted, sample size at registration, final sample size included in the DCE, type of DHI surveyed), DCE features (attribute development method, attributes identified, number of choice sets, number of attributes, number of levels, whether pretesting was conducted, whether and how a payment vehicle was defined), DCE administration (survey administration mode, whether an incentive was provided), and statistical analysis (regression models used, whether subgroup analysis was conducted, whether a monetary value and statistical uncertainty were reported).
The selection process of studies included in this review is presented in the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) flowchart. Data from the included studies were planned to be analysed descriptively, and presented in tables, graphs and charts, followed by a narrative synthesis describing how the results relate to the objectives of the review.
The search strategy yielded a total of 1,846 records. Among them, 1,319 were from Embase, 510 from PubMed, 14 from Scopus, and 3 were from Web of Science. 442 records were identified as duplicates, of which 409 were automatically identified, and 33 were manually identified. After removing duplicates, 1,404 records were put forward to title and abstract screening, from which 1,391 were excluded. The remaining 13 studies were assessed for eligibility in a full-text review. Seven studies were excluded because they did not have a DCE study design. Two studies were excluded because they were not focused on stroke, and the remaining four studies were excluded because they were abstracts only. No studies met the inclusion criteria of this review; therefore, none were included in the review. The results of the search and study selection process are presented in a PRISMA flowchart (Figure 1).
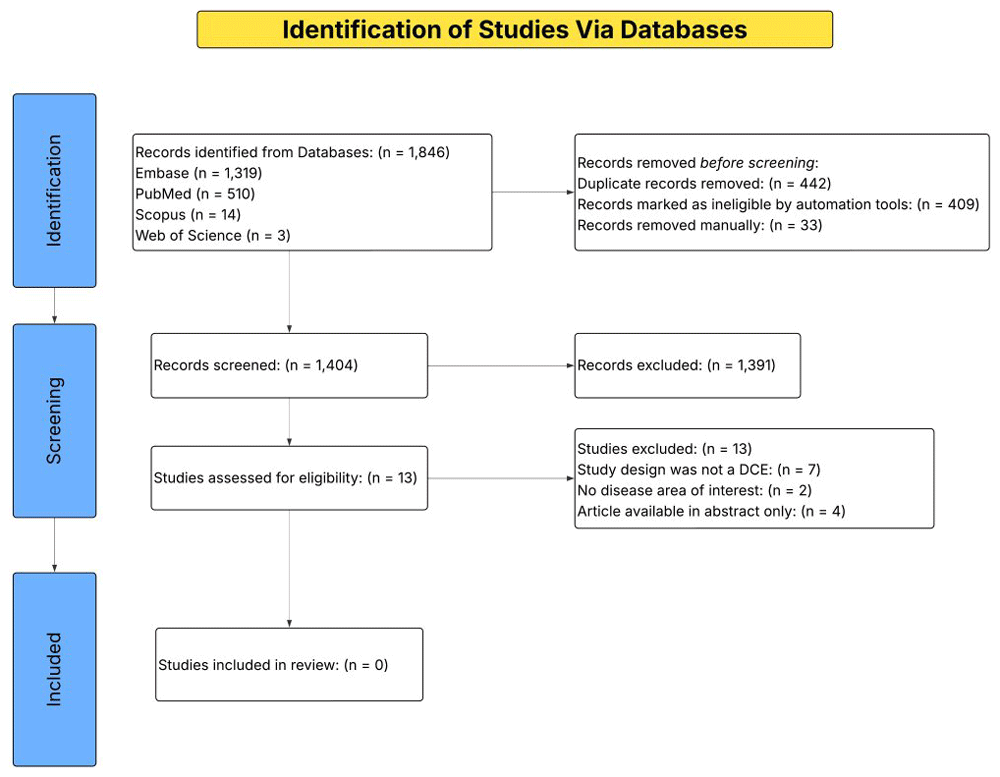
Figure 1 shows a PRISMA flowchart illustrating the search results obtained from Embase, PubMed, Scopus, and Web of Science searches. The flowchart also shows the duplicate articles removed, the number of articles that were put through screening, and those that were included in the review.
This scoping review aimed to identify the available evidence on the application of DCE methodology to assess preferences for DHIs in stroke care. Despite the increase in the use and application of DHIs in stroke care in particular (Hankinson et al., 2022; Ho et al., 2024; Salgueiro et al., 2022; Xu et al., 2023), the review did not find any study using DCE methodology to evaluate DHIs in stroke care. It is easy to conclude that this could be expected considering DCEs are cognitively demanding (Flynn et al., 2016) and that some types of strokes are associated with cognitive impairment (Nijsse et al., 2017). However, it should be noted that the same methodology has been applied successfully in other interventions in stroke care, including in eliciting preferences for exercise and post-stroke self-management programmes (Doshi et al., 2023; Geidl et al., 2018), and in eliciting preferences for community services that support life after stroke (Burton et al., 2014). Moreover, DCEs in stroke care target not only stroke survivors but also other stakeholders like family, relatives, friends, caregivers and healthcare professionals, among others.
To our knowledge, this review is the first of its kind to map existing studies applying DCE methodology in eliciting preferences for DHIs in stroke care. The application of rigorous and transparent methods for conducting a scoping review, as proposed by the JBI methodology, is advantageous. By registering the review’s protocol (with all the details of the methods to be used in carrying out this review), developing the search strategy in collaboration with an information specialist and involving multiple reviewers in the selection of studies, allows reproducibility and ensures reliability and validity of the study results.
Although the study inclusion criteria were broad (with no age or year of publication restrictions), the review may have missed some studies, as it focused on peer-reviewed studies published in the English language and available in full text. The exclusion of editorials, commentaries, and grey literature (such as dissertations, theses, conference proceedings, and preprints), as well as studies published in languages other than English, could have led to the omission of potentially relevant research. By excluding studies available only in abstract form, we may have overlooked emerging research that is yet to be published in full. We encountered 2 potentially relevant studies, however, one was available as an abstract only (Fischer & Mühlbacher, 2023, p. 15), while the other did not disaggregate its results by disease area, despite 50% of participants in the study being stroke patients (Laver et al., 2013)
The non-existence of DCE studies of DHIs in stroke care may be attributed to several factors. Firstly, many stroke survivors suffer from a range of impairments, including cognitive deficits, aphasia and mobility challenges (Mellon et al., 2015; Louie et al., 2022; Tsouli et al., 2009). These can hinder participation in complex research tasks involved in DCEs. For instance, people with aphasia face challenges in expressing their needs, as well as in reading and writing (Poirier et al., 2024). Hence, tasks that require reading complex information may be particularly challenging. It is no surprise, then, that several randomised controlled trials excluded stroke patients with aphasia, even though aphasia is common among stroke survivors (Vaughan & Manning, 2023). Regardless of the severity of the condition, stroke survivors also experience fatigue (Ingles et al., 1999), which impacts their activities of daily living, ranging from straightforward tasks such as eating and dressing to more demanding roles like meal preparation (Niyomyart et al., 2024). Stroke patients reported that visual and cognitive difficulties prevented them from accessing mHealth services (Papadopoulos-Nydam et al., 2021). Therefore, when designing DCEs of DHIs targeting stroke survivors, researchers need to adapt to these challenges by simplifying attributes and levels and consider using pictures instead of text.
Secondly, the use of proxy respondents in research introduces other challenges. Previous studies on surrogate decision-making in the context of stroke showed that patients' and surrogates' agreement on research scenarios was low, indicating that surrogates may be less reliable at eliciting patients' preferences (Bryant et al., 2013). Nevertheless, family members and caregivers play an integral role in decision-making during the time stroke survivors experience impairments (Bryant et al., 2013). Therefore, designing DCEs that capture patients' preferences through proxy respondents may be a viable option in such cases. However, the involvement of proxy respondents should be considered as a last resort, especially in cases where the DCE is designed to capture patient-centred preferences, ensuring that the results accurately reflect patients' priorities.
Furthermore, recruiting study participants from the primary target group in surveys is in general a challenging task. Even the proxy participants (family or caregivers) are often identified by the stroke survivors themselves. This problem is exacerbated by the lack of well-coordinated patient organisations, which impacts the existence of central registries with patients’ contact information. The existence of hospital registries does not guarantee a solution to this problem, as securing ethical approval for research studies is challenging in hospital settings.
Moreover, DCEs are complex techniques. The methodology is not widely recognised among decision-makers and implementation researchers; therefore, it is not highly demanded when seeking evidence to support decisions. Besides, stroke prevalence is higher among older adults (Feigin et al., 2025), a demographic group that faces significant challenges in using technology (Chen et al., 2023). Consequently, conducting DCEs of DHIs in stroke care may be less pertinent. However, with the increasing global incidence of stroke among the younger population (Feigin et al., 2024), a group that is adept with digital technologies, there is a need for DCEs to elicit preferences for DHI in stroke care.
Developing DCEs for DHIs also presents challenges. Researchers must decide how broad or specific the attributes and levels should be. For instance, developing attributes and levels for mHealth is difficult because this category includes a wide variety of DHIs, such as medication adherence applications, remote monitoring, teleconsultation, and telerehabilitation. Even within these examples, there are different DHIs, further complicating the process of developing attributes and levels. Additionally, rapid technological changes create another problem. By the time a DCE for a DHI is finalised, the technology involved may be outdated and the results be less relevant for decision making.
DCEs of DHIs in stroke care remain valuable and relevant because they provide evidence of user preference for DHIs in stroke care. Therefore, implementing this methodology in future studies of DHIs in stroke care will expand its development, ensuring greater inclusion, easier access, and more efficient resource utilisation.
The data for this article consists of bibliographic references, which are included in the references section.
Zenodo PRISMA-ScR checklist for “Application of discrete choice experiments to assess preferences for digital health interventions in stroke: a scoping review” https://zenodo.org/records/17350271 (Forde et al., 2025b).
This project contains the following reporting guidelines:
PRISMA-ScR checklist for “Application of discrete choice experiments to assess preferences for digital health interventions in stroke: a scoping review”
The reporting of the review was guided by the PRISMA-ScR checklist. This file is the completed checklist for this review. It outlines where each of the recommended reporting items is addressed within this review.
Data is available under the terms of the Creative Commons Attribution 4.0 International license (CC-BY-4.0).
Provide sufficient details of any financial or non-financial competing interests to enable users to assess whether your comments might lead a reasonable person to question your impartiality. Consider the following examples, but note that this is not an exhaustive list:
Sign up for content alerts and receive a weekly or monthly email with all newly published articles
Register with HRB Open Research
Already registered? Sign in
Submission to HRB Open Research is open to all HRB grantholders or people working on a HRB-funded/co-funded grant on or since 1 January 2017. Sign up for information about developments, publishing and publications from HRB Open Research.
We'll keep you updated on any major new updates to HRB Open Research
The email address should be the one you originally registered with F1000.
You registered with F1000 via Google, so we cannot reset your password.
To sign in, please click here.
If you still need help with your Google account password, please click here.
You registered with F1000 via Facebook, so we cannot reset your password.
To sign in, please click here.
If you still need help with your Facebook account password, please click here.
If your email address is registered with us, we will email you instructions to reset your password.
If you think you should have received this email but it has not arrived, please check your spam filters and/or contact for further assistance.
Comments on this article Comments (0)